
There are no FDA approved Drugs, Vaccines, or medicine for Coronavirus treatments till the date. However, different medicines are being used all over the world for the treatment of severe cases of COVID-19. Here we have listed the name of medicine, the science behind their work, and their effectiveness in COVID-19 treatments.
List of Medicine for Coronavirus (Covid-19)
- Remdesivir
- Hydroxychloroquine
- Favilavir
- Azithromycin
- Plasma Therapy
- Sepsivac (The Leprosy Drug)
- Tocilizumab
1. Remdesivir
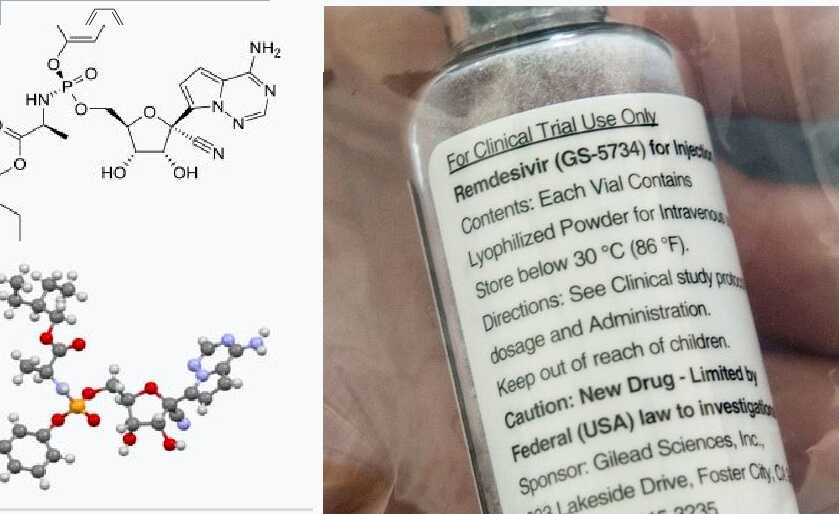
Remdesivir is originally an antiviral-medicine developed by company Gilead Sciences. The medicine was developed for Hepatitis C disease but was not that effective. It is an injection that is injected into the vein. This medicine has been used for emergency cases in India and the USA as medicine for Coronavirus (COVID-19) Treatments. Also, it is used in Japan and the UK from the Month of May 2020.
How Remdesivir works?
Remdesivir works when the enzyme replicating the genetic material for new virus generation accidentally grabs nucleoside analog (which is a mimic structure produced from the medicine) rather than the natural molecule. So the virus incorporates nucleoside from medicine into the growing RNA strand. So it blocks other RNA from replication and controls the number of replication of the virus.
The medicine also has side effects of Respiratory failure, Low RBC and Platelets, yellow skin, low albumin, and organ impairment.
The medicine was previously tested for other pandemics like Ebola and SARs, but it worked for non- of them. Although thousands of patients have been cured with this medicine, clinical trials are still on process. The medicine is now authorized to be given to the patient for a dose of 10 days.
The WHO has provided the license to manufacture Remdesivir for distribution to 127 countries. Also, the manufacturer can set their own price and should not pay the royalty.
2. Hydroxychloroquine

Hydroxychloroquine is another medicine that is being used as medicine for Coronavirus. It is known by the name Plaquenil. The medicine was originally used for the treatment of Malaria. It is taken by mouth and not the injection. The medicine works by blocking the toll-like receptors (TLR) on plasmacytoid dendritic cells (PDCs).
The most common side effects of this medicine are Muscle pain, weak in vision, vomit, headache, and heart problems.
EMA (European Medicine Agency) provided a statement on 29 May 2020 to closely monitor people who are receiving hydroxychloroquine for the treatment of Coronavirus (COVID- 19). Although it has been used in many countries, it has been banned in countries like France, Italy, and Belgium.
WHO suspended it from global drug trials from May 26, 2020, due to severe safety concerns. However, on June 3, 2020, WHO announced to resume its global trial after monitoring no risk of death.
3. Favilavir

Favilavir is China’s first approved medicine for Coronavirus used. Similar to Remdesivir it is an anti-viral drug. Over the past years, the medicine has been used for a number of diseases like Influenza, Ebola, Respiratory syncytial and Zika virus. The drug works by preventing the replication of the virus inside the organ.
Its trial is going on in Japan, China, and India. According to reports, the patients in Shenzhen some of the patients turned Coronavirus negative after using for four days of becoming positive.
However, the US Food and Drug Administration (FDA) has not regarded this medicine as an effective treatment of Coronavirus disease or COVID-19 Disease.
4. Azithromycin
Azithromycin also is known as Z-Pak is an antibiotic medicine. Although the drug is used to fight the bacteria, also it is known to have some anti-viral properties.
The drug is found effective under some trials in combination with hydroxychloroquine. The trials have reported that its use with HCQ had a more good response than using HCQ alone. The medicine has gone severe trials in countries like the USA by the University of Utah, Rutgers University, and Duke University.
However, the major side effect of the disease is heart side effects as it causes abnormal changes in the rhythm of the heart. So, the medicine is not that suitable for patients with heart diseases.
5. Plasma Therapy
Plasma Therapy involves the administration of blood plasma of patients who have recovered from the infection.
How does plasma therapy work for COVID- 19 Treatment?
The real concept about this medicinal process is that the blood of recovered patients contains anti-bodies in virus against blood.
Many trials are going on in New- Delhi India for the effectiveness of this therapy against the treatment of COVID-19.
According to the science of the Immune System, the body sends out a defensive cell called Arm of Cells. This cell including white blood cells stays in the liquid portion of cells called plasma. The survived patient is believed to develop a large number of antibodies in their plasma. So these plasma rich blood cells will help to fight the coronavirus disease, positive patient.
6. Sepsivac (The Leprosy Drug)
Sepsivac is a medicine that has been used since 1966 for the treatment of leprosy in India. The drug was used for treating four patients in Chandigarh. And now it is in the third phase of the trial with 4500 people.
How Sepsivac- Leprosy drug works?
It contains heat-killed Mycobacterium W (Mw) which is an immunomodulator. So Sepsivac modifies the immune system and helps in more antibody formation.
7. Tocilizumab
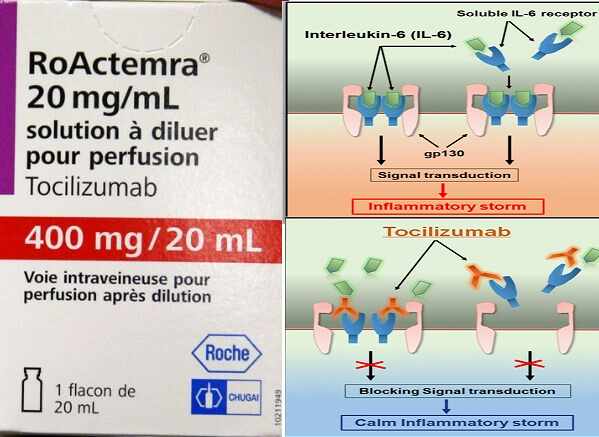
Tocilizumab medicine is also known for IL-6 Inhibitor property. The IL-6 helps to control the Inflammatory Storm. The Inflammatory storm refers to an excessive inflammation that is out of control.
It is also an Immuno-Modulator medicine. Meaning that it also helps to strengthen the immune system of the body and helps in growing antibody mechanism. Although medicine is found useful, it is not FDA approved.
Above mentioned are the medicine that is being used for the treatment of COVID-19 under sever-cases. However, the FDA has not approved any medication for the treatment of Coronavirus. The trial is set in many countries all over the world and is in different phases.
